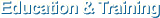- question1.Will agreement become effective only after IRB’s authorization?
-
- answer Agreement becomes effective regardless of IRB authorization period. However, it takes time to review protocol, check e-CRF item and produce it into website that consultation from initial research stage is highly suggested. Clinical research although must be progressed after IRB’s authorization.
-
- question2. Would there be any other thing required to use eVelos System such as to install separate program?
- answer No additional program is required to be installed and eVelos System is available in the computer with internet access.
- question3. How can eVelos System access and log-in error to use e-CRF be solved?
- answerIn order to log-in to eVelos System, connect to http://eresearch.ncc.re.kr, and enter user name (ID) and password. Check if URL of operation version is correct and remember that ID and passwords are case sensitive
Following explains how to deal with error message during log-in.- [Access Details]
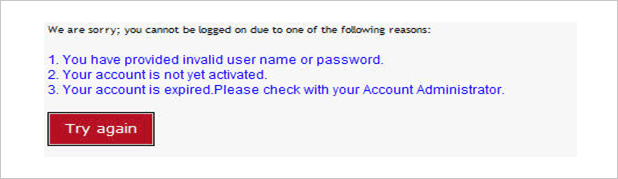
- [Account Block]
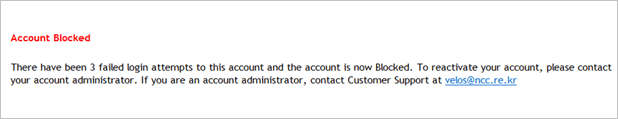
- [Session Management]
- [Pop-up Blocker]
-
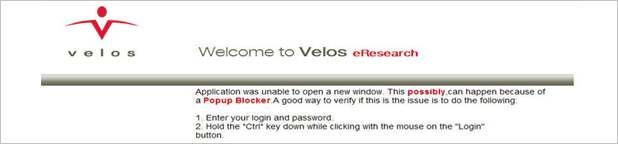
If Pop-up blocker is in use, following windows will display and log-in won’t work. Try one of followings to resolve the error.
(1) Disable the pop-up blocker from Tool > Pop-up blocker from menu bar of internet browser
(2) Permit eResearch.ncc.re.kr URL from Tool > Pop-up blocker > Pop-up blocker setup
- answerIn order to log-in to eVelos System, connect to http://eresearch.ncc.re.kr, and enter user name (ID) and password. Check if URL of operation version is correct and remember that ID and passwords are case sensitive
- question4.The first patient is not registered in the system. What should be checked before registering the first patient?
- answer The first patient is registered only when Protocol Management > Study is set to Active/Enrolling. You only can update once before enrolling the first patient per organization. Please refer to Protocol Management from system training for the detail and method.
- question5. Menu (Manage Patient > New) for patient registration and New button for creating e-CRF are not shown in the system. What are the criteria to access to some menus and what’s required to the patient enrollment and e-CRF preparation?
- answer Data Management Team controls the access by the group which is categorized by role during study (Investigator, CRC, Monitor, DM, and etc.) If user ID is registered under investigator group, he or she is not permitted to enroll/register the patient and create e-CRF. Investigators may have additional access to data entry provided that certain training has been completed. When filling in e-Velos registration request, fill PI+CRC in permissions required field, such data entry access will be given.